Abstract
Background: CTCLs are generally incurable, and current systemic therapies seldom provide high rates of durable responses, with overall response rates of 24-68% (Zinzani et al, Crit Rev Oncol Hematol, 2016), and median time to next therapy (TTNT), a marker of disease control and meaningful endpoint for clinicians/patients (pts), of 3.9 months (Hughes et al, Blood, 2015). The phase 3 ALCANZA study (Prince et al, Lancet, 2017; NCT01578499) was the first randomized study of a new systemic drug compared to standard systemic therapy, and the largest reported phase 3 trial, in CTCL pts. At the initial report, after a median follow-up of 22.9 months (data cut-off May 31, 2016), ALCANZA showed a significantly higher rate of durable responses (per independent review facility assessment) with the CD30-directed antibody-drug conjugate brentuximab vedotin vs physician's choice (PC: methotrexate or bexarotene) for CD30+ CTCL. Here, we provide previously unreported and updated analyses per investigator (INV) assessment from ALCANZA, after a median follow-up of 33.9 months (data cut-off June 30, 2017), further evaluating treatment response and clinical benefit in CTCL pts.
Methods: Adults with previously treated CD30+ mycosis fungoides (MF) or primary cutaneous anaplastic large cell lymphoma (pcALCL) requiring systemic therapy were randomized to brentuximab vedotin 1.8 mg/kg IV Q3W (up to 16 cycles) or PC (up to 48 weeks: methotrexate 5-50 mg PO QW or bexarotene 300 mg/m² PO QD). Primary endpoint was rate of objective response lasting ≥4 months (ORR4). Key secondary endpoints were complete response (CR) rate, progression-free survival (PFS), and symptom burden measured by the symptom domain of the Skindex-29 quality-of-life (QoL) tool. TTNT was defined as the time from randomization to the date of the first documentation of antineoplastic therapy or last contact date for pts who never took antineoplastic therapy.
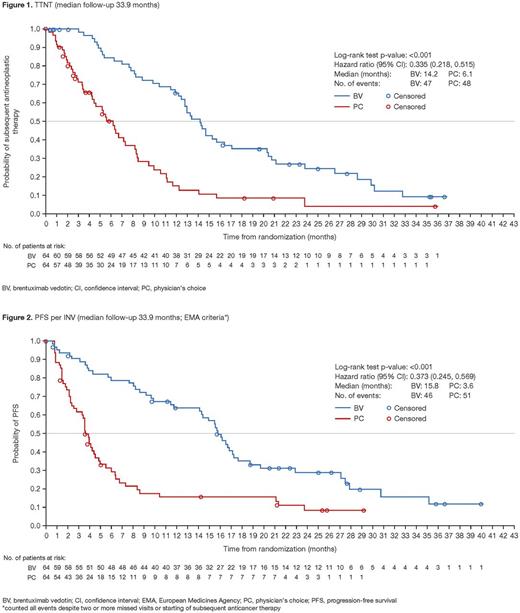
Results: 131 pts were randomized, with 128 pts in the intent-to-treat population (97 MF, 31 pcALCL; 3 excluded for insufficient CD30 expression) assigned to brentuximab vedotin (n=64; median CD30 expression 32.5%) or PC (n=64; median CD30 expression 31.3%). Longer-term data per INV assessment continued to show significant clinical benefit for brentuximab vedotin vs PC. At last follow-up, in the brentuximab vedotin and PC arms, 73% and 75% of pts had received subsequent antineoplastic therapy, respectively; TTNT was significantly longer with brentuximab vedotin vs PC (median 14.2 vs 6.1 months; HR 0.335; 95% CI, 0.218-0.515; p<0.001) (Fig 1). In the brentuximab vedotin arm, the probability of pts not requiring subsequent antineoplastic therapy was 65.5% at 1 year and 24.6% at 2 years. Improved long-term disease control observed with brentuximab vedotin vs PC was consistent with other efficacy improvements as demonstrated by an INV-assessed ORR4 of 60.9% vs 7.8% (p<0.001), CR rate of 18.8% vs 0% (p<0.001), and median PFS of 15.8 vs 3.6 months (HR 0.373; 95% CI, 0.245-0.569; p<0.001) (Fig 2) with brentuximab vedotin vs PC. In addition, QoL assessed by Skindex-29 showed significantly greater symptom reduction for brentuximab vedotin vs PC (mean maximum reduction -28.08 vs -8.62; p<0.001). Peripheral neuropathy (PN), a known toxicity, was the most commonly reported adverse event with brentuximab vedotin. Longer-term PN data showed that 86% (38/44) of pts in the brentuximab vedotin arm had complete resolution (26/44) or improvement (≥1 grade; 12/44) of all PN events, compared with 82% (36/44) in the original analysis. PN improved over time; at last follow-up, in brentuximab vedotin treated pts, ongoing PN was Grade 1/2 in 15/3 pts (no Grade 3/4 PN) compared with Grade 1/2 in 17/5 pts in the original analysis.
Conclusions: Longer-term analyses from ALCANZA continue to provide compelling evidence of improved response rates, PFS, and QoL with brentuximab vedotin vs PC in CD30+ CTCL. Grade 1-2 PN persists in 28% (18/64) of pts. We present new TTNT data and show an extended TTNT with brentuximab vedotin compared with PC suggesting that single-agent treatment with brentuximab vedotin not only provides high rates of response and symptom control, but also leads to durable and clinically meaningful responses in this population with relapsed/refractory disease. These data further support integration of brentuximab vedotin into the standard management of CD30+ CTCL pts who require systemic therapy.
†Joint lead author
Horwitz: Mundipharma: Consultancy; HUYA: Consultancy; BMS: Consultancy; Forty-Seven: Consultancy, Research Funding; Millennium Pharmaceuticals, Inc.: Consultancy; Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited: Research Funding; Infinity/Verastem: Consultancy, Research Funding; Kyowa Hakko Kirin: Consultancy, Research Funding; Spectrum Pharmaceuticals: Research Funding; Seattle Genetics: Consultancy, Research Funding; Aileron Therapeutics: Research Funding; Celgene: Consultancy, Research Funding; ADCT Therapeutics: Research Funding. Scarisbrick: NHS: Employment; Millennium Pharmaceuticals, Inc.: Consultancy, Honoraria; Innate Pharma: Consultancy; Mallinckrodt: Consultancy, Honoraria; Actelion: Consultancy. Dummer: Millennium Pharmaceuticals, Inc.: Consultancy, Honoraria. Duvic: Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited: Membership on an entity's Board of Directors or advisory committees, Other: Research funding (paid to institution); . Kim: Eisai: Membership on an entity's Board of Directors or advisory committees, Research Funding; Forty Seven Inc: Membership on an entity's Board of Directors or advisory committees, Research Funding; Horizon Pharma: Consultancy, Research Funding; Innate Pharma: Consultancy, Research Funding; Kyowa-Kirin-Pharma: Membership on an entity's Board of Directors or advisory committees, Research Funding; Medivir: Membership on an entity's Board of Directors or advisory committees; Merck: Research Funding; miRagen: Research Funding; Neumedicine: Research Funding; Portola: Consultancy, Research Funding; Seattle Genetics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Soligenix: Research Funding; Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited: Research Funding; Tetralogic: Research Funding; Millennium Pharmaceuticals, Inc.: Membership on an entity's Board of Directors or advisory committees. Walewski: Millennium Pharmaceuticals, Inc.: Consultancy, Honoraria. Whittaker: Millennium Pharmaceuticals, Inc.: Membership on an entity's Board of Directors or advisory committees; Galderma: Research Funding; Seattle Genetics: Other: Investigator in a clinical trial; Celgene: Honoraria; Innate Pharma: Other: Investigator in a clinical trial. Zinzani: Celgene, Janssen, Gilead, Roche, Takeda, BMS, MSD, Sandoz, Servier, Mundipharma: Speakers Bureau; Celgene, Roche, Janssen, Gilead, Takeda, BMS, MSD, Servier, Sandoz, Mundipharma: Honoraria; Merck: Consultancy, Other: Advisory board. Eradat: Pharmacyclics: Consultancy, Research Funding; Novartis: Research Funding; Gilead: Consultancy, Honoraria, Research Funding, Speakers Bureau; Genentech: Consultancy, Honoraria, Research Funding, Speakers Bureau; Roche: Consultancy, Research Funding; Abbvie: Consultancy, Honoraria, Research Funding, Speakers Bureau; Celgene: Research Funding. Sanches: Leo Pharma: Membership on an entity's Board of Directors or advisory committees. Ortiz-Romero: Millennium Pharmaceuticals, Inc.: Consultancy; Abbvie: Other: Travel expenses; Janssen: Other: Travel expenses; Actelion: Consultancy; Innate Pharma: Consultancy; MEDA: Research Funding; 4SC: Consultancy; Patent of PLG1 mutation for diagnostic or treatment of cutaneous lymphomas: Other: Patent of PLG1 mutation for diagnostic or treatment of cutaneous lymphomas; Roche: Other: Travel expenses. Akilov: Trillium Therapeutics: Research Funding; Medivir: Consultancy; Actelion Pharmacuticals: Consultancy, Research Funding; Seattle Genetics: Consultancy. Trotman: Roche: Membership on an entity's Board of Directors or advisory committees, Other: All positions non-remunerated, Speakers Bureau; Celgene: Membership on an entity's Board of Directors or advisory committees, Other: Funding facilitating research paid to third parties. All positions non-remunerated; Takeda: Membership on an entity's Board of Directors or advisory committees, Other: Position non-remunerated; Janssen Cilag: Membership on an entity's Board of Directors or advisory committees, Other: Funding facilitating research paid to third parties. All positions non-remunerated, Speakers Bureau. Dalle: Kyowa Hakko Kirin Pharmaceutical: Research Funding. Weichenthal: Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited: Research Funding; Takeda Oncology: Consultancy, Other: Travel expenses; TEVA Pharmaceuticals: Consultancy, Other: Travel expenses; Millennium Pharmaceuticals, Inc.: Consultancy. Fisher: Seattle Genetics Inc.: Membership on an entity's Board of Directors or advisory committees. Stadler: Millennium Pharmaceuticals Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees; Seattle Genetics Inc.: Consultancy; 4SC: Consultancy; ICN: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Roche: Membership on an entity's Board of Directors or advisory committees; Johnson and Johnson: Membership on an entity's Board of Directors or advisory committees. Feldman: AbbVie: Speakers Bureau; Janssen: Speakers Bureau; Seattle Genetics: Honoraria, Research Funding, Speakers Bureau; Bristol-Myers Squibb: Consultancy; Pharmacyclics: Speakers Bureau; Celgene: Speakers Bureau; Kite Pharma: Speakers Bureau. Kuzel: Emmaus: Membership on an entity's Board of Directors or advisory committees; Eisai: Data monitoring committee honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; AbbVie: Membership on an entity's Board of Directors or advisory committees; Exelexis: Membership on an entity's Board of Directors or advisory committees; Astellas: Honoraria; Merck: Other: Data monitoring committee honoraria; Bayer: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Honoraria, Research Funding; Amgen: Other: Data monitoring committee honoraria; Seattle Genetics Inc.: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Sanofi: Honoraria; Genentech: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Medivation: Honoraria; Celgene: Honoraria; Argos: Other: Data monitoring committee honoraria. Gru: Seattle Genetics: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Consultancy, Membership on an entity's Board of Directors or advisory committees. Wang: Seattle Genetics: Employment, Equity Ownership. Palanca-Wessels: Seattle Genetics: Employment, Equity Ownership. Lin: Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited: Employment. Liu: Takeda Pharmaceutical Company Limited: Equity Ownership; Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited: Employment. Little: Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited: Employment; Takeda Pharmaceutical Company Limited: Equity Ownership. Prince: Millennium Pharmaceuticals, Inc., a wholly owned subsidiary of Takeda Pharmaceutical Company Limited: Research Funding; Millennium Pharmaceuticals, Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene Corporation: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Eisai: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal